6V Micro Water Pump | Food Grade & RoHS Certified | PINMOTOR
Customized Service
To provide customers with quality products and satisfactory service
PYRP310-XA
Tiny Water Pump
-
Contamination-Free Design: Oil-free diaphragm structure ensures zero fluid contamination, meeting RoHS & REACH standards for laboratory use.
-
Corrosion Resistance: Optimized for handling medical-grade alcohol, gels, and weak acid/alkali reagents.
-
Precision Pulse Control: Fine-tuned internal valve geometry minimizes fluid pulsation, ensuring consistent spray or droplet volume.

Product Information
|
PYRP310-XA Tiny water pump |
||||
| *Other Parameters: according to customer demand for design | ||||
| Rate Voltage | DC 3V | DC 3.7V | DC 4.5V | DC 6V |
| Rate Current | ≤1000mA | ≤810mA | ≤660mA | ≤500mA |
| Power | 3.0w | 3.0w | 3.0w | 3.0w |
| Air Tap O.D. | φ 5.0mm | |||
| Water Flow | 50-200 mLPM | |||
| Max Maximum Pressure | ≤-20Kpa (-150mmHg) | |||
| Noise Level | ≤65db (30cm away) | |||
| Life Test | ≥100,000 Times (ON 2S,OFF 2S) | |||
| Pump Head | ≥0.5m | |||
| Suction Head | ≥0.5m | |||
| Weight | 42g | |||
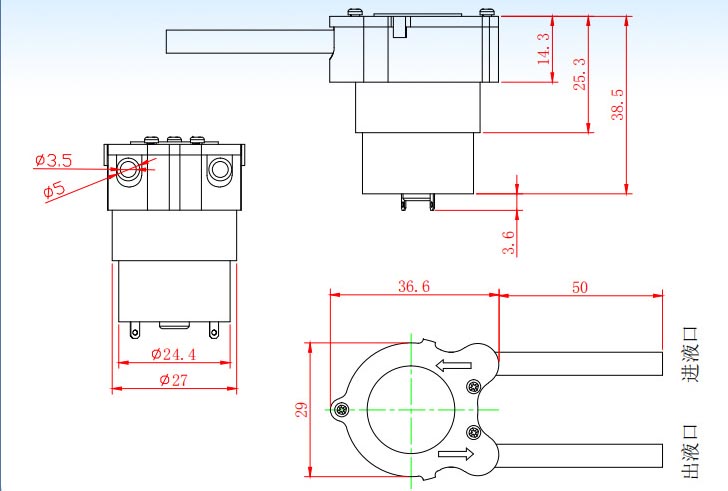
Specification Engineering Drawing
Images for micro gear pump---100% live-action shooting, Quality Guarantee

Manufacturing Quality & Compliance
At Pinmotor, we implement rigorous quality control to ensure our pumps integrate seamlessly into your high-volume production lines. The PYFP310-XG series is manufactured in our automated facility, complying with the following global medical, automotive, and environmental standards:

ISO 9001:2015 Certified
Our comprehensive quality management system ensures high-precision performance for every 310 series micro pump. From stringent raw material inspection to the 100% final functional testing of every unit, we guarantee consistent flow rates and pressure stability. Each pump undergoes rigorous leakage and motor-load calibration to meet the demanding reliability standards of medical disinfection and laboratory integration.

IATF 16949 Compliant
We leverage automotive-grade process discipline (IATF 16949 standards) to guarantee ultra-low defect rates and long-term reliability for medical and professional hygiene applications. By applying rigorous stress-testing and precision assembly to our 310 micro pump series, we ensure that every unit delivers consistent, maintenance-free performance in demanding, high-frequency environments.

RoHS & REACH Fully Compliant
Every component in the 310G series—from the high-durability motor brushes to the medical-grade diaphragm and reinforced housing—is strictly RoHS and REACH compliant. Our commitment to hazardous-substance-free manufacturing ensures your end-product meets the most stringent environmental regulations for the EU and North American markets, including California Prop 65 standards. We provide full material traceability to simplify your certification process and guarantee consumer safety.
Technical Note: Full documentation, including material safety data sheets (MSDS) and compliance certificates, is available upon request for your engineering audit.
Application
| Industry Sector | Specific Applications | Technical Integration Benefit |
| Medical & Healthcare | IVD (In Vitro Diagnostic) analyzers, Clinical chemistry buffers, Automated hand sanitizers. | Chemical resistance: Handles alcohol and reagents without degradation. |
| Laboratory Automation | Titration systems, Liquid handling robots, Reagent dosing modules. | Precision control: Consistent flow rates for accurate laboratory measurements. |
| Hygienic Appliances | Touchless soap dispensers, Wall-mounted disinfectant sprayers, Dental irrigation tools. | Quiet operation: Low-noise (<40.5 dB) profile suitable for quiet indoor environments. |
| Industrial Integration | Inkjet printer ink circulation, Lubrication micro-systems, Cooling for small electronics. | Self-priming: Reliable 0.5m suction lift for flexible internal component layout. |
| Consumer Wellness | High-end aromatherapy diffusers, Facial misting devices, Smart hydration systems. | Food-grade safety: Compliance with FDA/RoHS standards for consumer contact. |

dishwasher

Electric decanter

Foam hand sanitizer

Water dispenser

Vacuum packing machine

Tea table
Best Micro Water Pump Manufacturer and Exporter In China
We can provide the best price and technical support for commercial projects.
Frequently Asked Questions: 310 Medical Micro Water Pump
What makes the 310 micro pump “medical-grade” compared to standard pumps?
The primary difference lies in material safety and reliability. Our medical-grade 310 pumps utilize FDA-compliant EPDM diaphragms and USP Class VI-compatible housings. Unlike consumer-grade pumps, these are manufactured in controlled environments to prevent contamination and undergo rigorous 500+ hour continuous duty cycle testing to ensure zero-failure performance in critical healthcare applications.
Can this micro pump handle high-viscosity medical gels or only thin liquids?
It is specifically engineered for both. While standard pumps often clog with thick fluids, our 310 series features a reinforced valve structure and a high-torque DC motor. This allows it to maintain a consistent flow rate for high-viscosity hand sanitizers and ultrasound gels without overheating or motor stall, ensuring a smooth, drip-free user experience.
How does this pump manage noise levels for hospital or bedside use?
Patient comfort is a priority. We’ve optimized the internal motor balance and used vibration-dampening elastomers to keep noise levels below 40.5 dB. In a typical medical environment, this is whisper-quiet. For integration, we recommend using flexible silicone tubing to further isolate mechanical resonance from your device’s housing.
Is the 310 micro pump compatible with alcohol-based disinfectants?
Yes. The pump head and internal seals are made from chemically resistant EPDM and PP polymers. These materials are specifically tested for long-term exposure to 75% Ethyl Alcohol and Isopropyl Alcohol (IPA). They won’t swell, crack, or degrade, which prevents internal leaks and maintains the chemical purity of your disinfectant.
What is the self-priming capability of this 6V micro liquid pump?
Our 310 diaphragm pump is fully self-priming up to 0.5 meters (1.6 feet). This means it can draw liquid from a reservoir located below the pump level without needing manual priming or a gravity feed. For medical designers, this provides greater flexibility when arranging internal components in compact, handheld, or wall-mounted devices.










